For millions of people with sleep apnea, a CPAP machine is a nightly necessity, a trusted medical device that helps them breathe easier and get restorative sleep. Relying on this equipment creates a fundamental expectation of safety.
However, that trust was shaken for many when it was discovered that the sound-dampening foam in millions of Philips Respironics CPAP, BiPAP, and ventilator devices could break down, leading to serious health issues. The revelation of the CPAP machine foam linked to cancer risk has left many families concerned, confused, and searching for answers about their health and legal options.
If you or a loved one used one of these recalled devices and later received a cancer diagnosis, it’s important to understand the situation and the steps being taken to hold the manufacturer accountable for the harm caused.
Key Takeaways about CPAP Machine Foam and Cancer Risk
- Millions of Philips CPAP, BiPAP, and ventilator devices were recalled due to the breakdown of their sound-abatement foam.
- The polyester-based polyurethane (PE-PUR) foam can degrade into particles or release harmful gases that users may inhale or ingest.
- The U.S. Food and Drug Administration (FDA) has classified this as a Class I recall, its most serious level, indicating a risk of life-threatening injury.
- Studies and adverse event reports have suggested a connection between the degraded foam and an increased risk of developing certain types of cancer.
- A multidistrict litigation (MDL) has consolidated thousands of personal injury lawsuits against Philips to streamline the legal process.
- Recent developments include a proposed settlement to resolve thousands of cancer-related claims from affected users.
What Caused the Philips CPAP Recall?
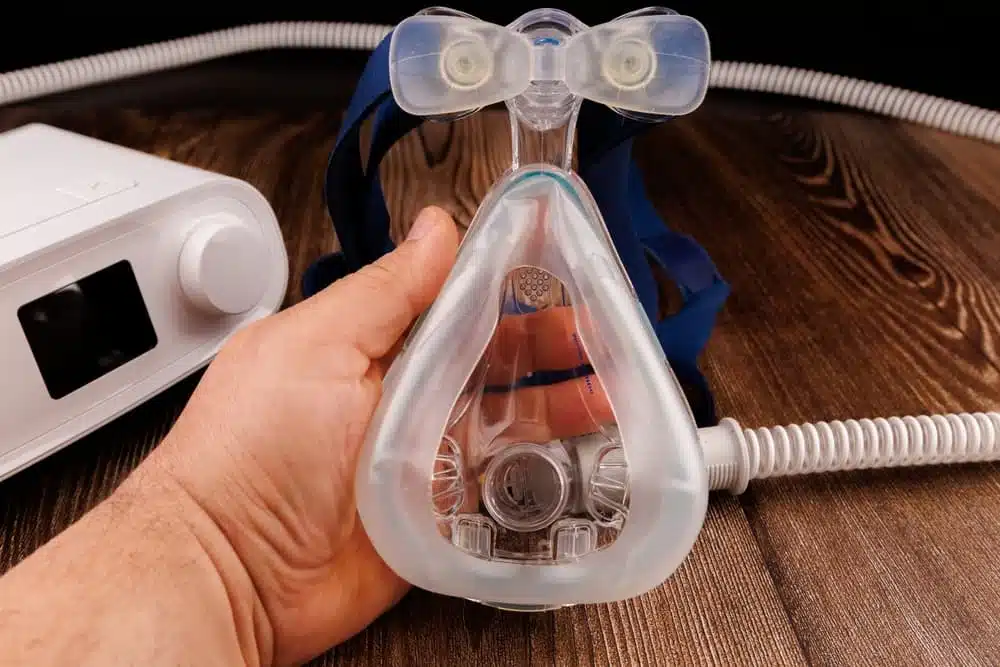
The core of the problem lies within the design of millions of Philips devices. To make them run more quietly, the manufacturer used a specific type of foam called polyester-based polyurethane (PE-PUR) for sound abatement—which is a way of reducing noise. Over time, particularly in warm or humid conditions, this foam can degrade.
This degradation process is dangerous for two primary reasons:
- Particle Inhalation: The foam can break down into tiny black particles that may travel through the machine’s air tubes and into a user’s lungs, airways, and digestive tract.
- Chemical Off-Gassing: The foam can also release certain harmful chemicals as gases, a process known as “off-gassing.” Users can then breathe in these toxic gases throughout the night.
Because of these dangers, the U.S. Food and Drug Administration (FDA) announced a sweeping recall of the affected devices. The recall was designated as Class I, which the agency reserves for situations where there is a reasonable probability that using the product will cause serious adverse health consequences or death. This classification highlights the gravity of the potential harm associated with the degrading foam.
Understanding the CPAP Machine Foam Linked to Cancer Risk
The chemicals released by the degrading PE-PUR foam are known as volatile organic compounds (VOCs). Some of these chemicals are considered carcinogens, which are substances capable of causing cancer in living tissue. When users breathe in these particles or gases night after night, their long-term exposure can increase their risk of developing serious health problems.
Medical research and a growing number of reports from patients have pointed toward a potential connection between using a recalled Philips CPAP machine and a subsequent cancer diagnosis. The continuous exposure to carcinogenic materials has been associated with a higher rate of certain cancers compared to individuals who used non-recalled devices.
Cancers Potentially Associated with Recalled CPAP Devices
While research is ongoing, many lawsuits allege that exposure to the PE-PUR foam particles and gases has contributed to the development of various cancers. Some of the cancers that have been identified in claims include:
- Lung Cancer
- Liver Cancer
- Kidney Cancer
- Bladder Cancer
- Brain Cancer
- Leukemia
- Non-Hodgkin Lymphoma
- Rectal Cancer
- Breast Cancer
- Prostate Cancer
The presence of these life-altering illnesses has formed the basis of thousands of personal injury claims filed against the manufacturer. These legal actions seek to hold the company responsible for allegedly failing to warn consumers about the dangers of the PE-PUR foam.
The Legal Response: What Is a Multidistrict Litigation (MDL)?
When a single product harms a large number of people across the country, handling each lawsuit individually would be incredibly inefficient for the court system. To solve this, the federal court system uses a process called Multidistrict Litigation (MDL).
An MDL consolidates similar civil cases from different federal districts into one single district court. In the case of the Philips recall, all federal lawsuits related to injuries from the CPAP machines have been transferred to the U.S. District Court for the Western District of Pennsylvania.
Here’s what an MDL does:
- Streamlines Pre-Trial Proceedings: A single judge oversees all the shared legal processes, like discovery (the formal process of exchanging information between the parties about the witnesses and evidence) and pre-trial rulings. This avoids duplicative work and conflicting rulings from different judges.
- Promotes Efficiency: It saves time and resources for everyone involved—the plaintiffs, the defendant, and the courts.
- Encourages Settlement: By bringing all parties to the table, an MDL often makes it easier to negotiate a global settlement that can resolve many of a defendant’s outstanding claims at once.
It is important to understand that an MDL is not a class-action lawsuit. In an MDL, each plaintiff’s lawsuit remains individual. If a settlement isn’t reached, any unresolved cases can be sent back to their original courts for trial. This structure allows for the efficiency of a combined process while still protecting each person’s right to have their specific case heard.
Key Developments in the Philips CPAP Lawsuit

The Philips CPAP MDL has been active for several years, with tens of thousands of claims filed. The number of adverse events reported to the FDA has been significant, with reports linking the foam breakdown to hundreds of deaths and tens of thousands of medical incidents. These reports have put additional pressure on the company to address the claims.
Recent progress in the litigation has provided a path forward for many injured individuals.
- Bellwether Trials: To test legal arguments and see how juries might react, a few representative cases, known as bellwether trials, are selected to be tried first. A significant bellwether trial resulted in a multi-million dollar verdict for a plaintiff who developed lung cancer, finding that Philips was liable for failing to warn users about the foam’s dangers.
- Proposed Cancer Settlement: Philips has proposed a substantial settlement fund intended to resolve a large number of the personal injury claims from users who developed cancer after using the recalled devices.
- Ongoing Cases: While a settlement may resolve many claims, the litigation is not over. Individual cases involving severe injury or death may continue, and trials for any unresolved claims are expected to move forward.
This proposed global settlement still requires approval from the court and the plaintiffs involved. It represents a major step toward providing financial relief to those who have suffered the most severe consequences of the device’s defects.
What Compensation Could Be Available in a CPAP Lawsuit?
While no amount of money can reverse a cancer diagnosis or undo the pain it causes, a legal claim can help secure financial stability for families facing immense challenges. The goal of a personal injury lawsuit is to recover compensation, often referred to as damages, for the losses a person has experienced due to another party’s negligence.
In the context of the CPAP lawsuits, compensation may be sought for several types of damages:
- Medical Expenses: This can include the costs of cancer treatments, surgeries, hospital stays, prescription medications, rehabilitation, and any future medical care that may be required.
- Lost Wages: If a person’s illness prevents them from working, they may be able to recover compensation for the income they have lost and are expected to lose in the future. This is sometimes called “loss of earning capacity.”
- Pain and Suffering: This is a legal term for the non-economic harm a person endures. It accounts for the physical pain, emotional distress, and loss of quality of life resulting from the injury and diagnosis.
- Wrongful Death: In the tragic event that a person passes away due to a cancer linked to a recalled CPAP device, their surviving family members may be able to file a wrongful death claim to seek compensation for their loss.
Each person’s situation is unique, and the potential compensation available will depend on the specific details of their case, including the severity of their illness and the financial impact it has had on their life.
What to Do If You Used a Recalled Philips Device

If you or a family member used a recalled Philips CPAP, BiPAP, or ventilator and are concerned about your health, there are a few practical steps you can consider taking. This information is for general awareness and is not a substitute for medical or legal guidance.
- Consult Your Doctor: Your health is the top priority. Speak with your physician about your use of a recalled device and any health concerns you have. They can advise you on the potential risks, recommend health screenings, and discuss safer alternative treatments for your sleep apnea. Do not stop or change your prescribed therapy without first talking to your doctor.
- Preserve the Device: If possible, do not discard the recalled machine. It may serve as important evidence in a potential legal claim. Follow the manufacturer’s and FDA’s instructions regarding the device and any replacement programs.
- Keep Detailed Records: Document everything related to your medical journey. This includes the dates of your cancer diagnosis, a list of all treatments you have received, and receipts for all related medical bills and out-of-pocket expenses. It can also be helpful to keep a journal detailing how your illness has affected your daily life.
Gathering this information can help create a clear picture of how you and your family have been affected by the recalled device, which is an important part of exploring your options for accountability.
Cancer and CPAP Machine Foam FAQs
Here are answers to some common questions that people have about the health risks and legal actions related to recalled Philips CPAP machines.
Find Out How We Can Help
When a medical device that was supposed to help you ends up causing serious harm, it’s a profound breach of trust. At Ferrer Poirot Feller, we have been holding corporations accountable for the harm their products cause for over 40 years. Our experience is rooted in standing up for people who have been injured by the negligence of large companies nationwide. We believe that forcing manufacturers to prioritize safety over profits can make a difference for everyone.
If you or a loved one used a recalled Philips CPAP device and were later diagnosed with cancer, you deserve to have your story heard. We are here to listen and discuss your situation. Contact our team at (214) 521-4412 or through our online form for a free and confidential case assessment. We handle cases on a contingency fee basis, which means you will not owe us anything unless we successfully recover compensation for you. Let us put our knowledge and resources to work for you.